Choosing an incentive spirometer: What matters for post-surgical lung recovery
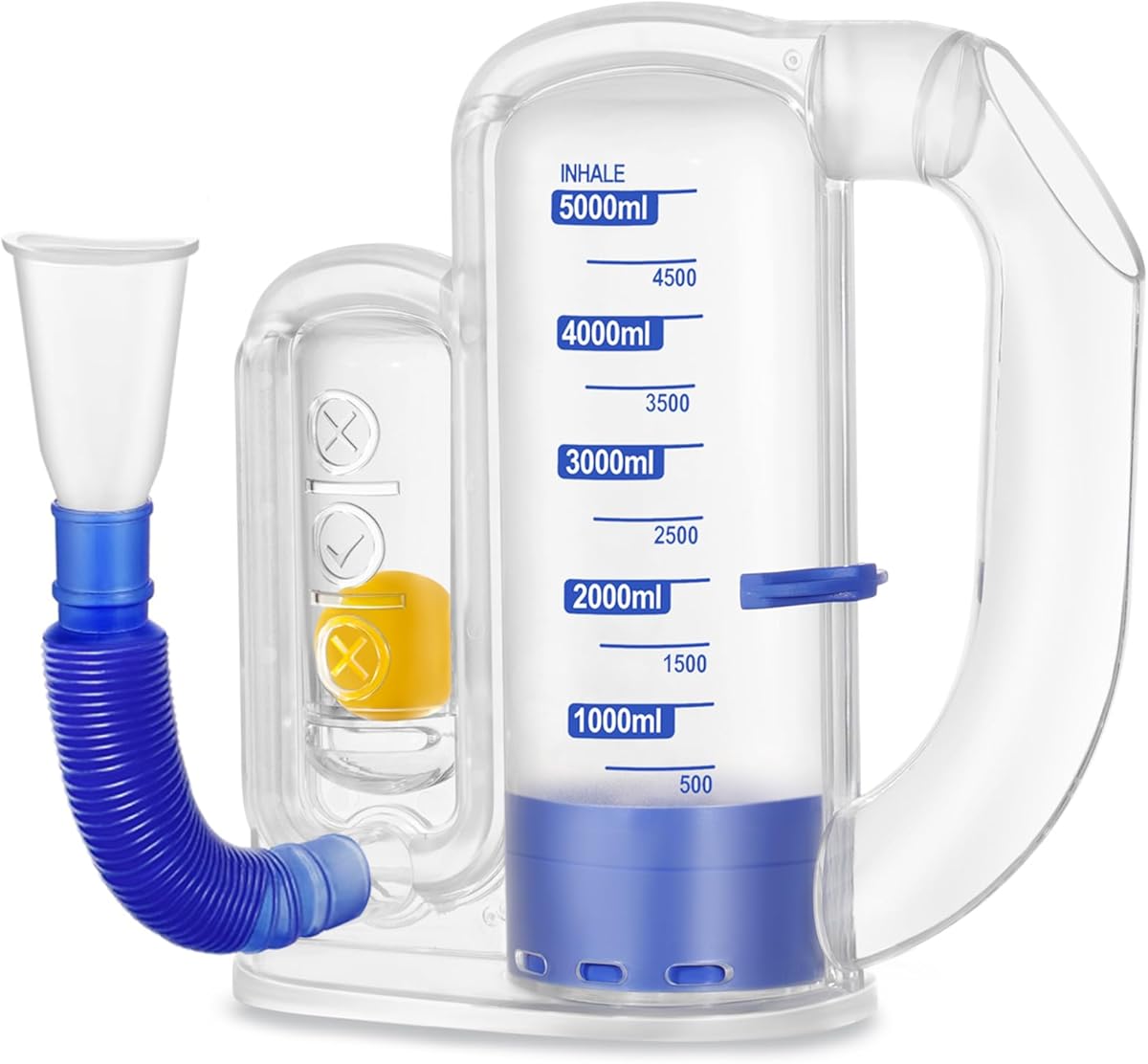
Incentive spirometers are medical devices designed to prevent pulmonary complications after surgery by encouraging deep breathing. This specific model functions as a flow-oriented device, measuring how quickly you can inhale to raise balls in chambers, which differs from volume-oriented models that track total lung capacity. Understanding this distinction is crucial for matching the device to your prescribed therapy.
Key Considerations Before Buying
- Flow vs. Volume Measurement: This product uses a flow-oriented design with floating balls, ideal for measuring inspiratory flow rate rather than total lung volume, which is often prescribed for post-operative patients to prevent atelectasis.
- Target Indicator Clarity: Look for clear, stable goal indicators—this model uses three separate chambers with colored balls, allowing you to set and visualize incremental breathing targets during recovery.
- Hygiene and Maintenance: Since these devices are used during vulnerable recovery periods, assess if components like the mouthpiece and tubing are detachable for cleaning, and if replacement parts are available.
What Our Analysts Recommend
A quality incentive spirometer should have durable, medical-grade plastic construction with no sharp edges, a smooth inhalation pathway with minimal resistance, and clear, easy-to-read measurement indicators. The device should maintain calibration over time, as inconsistent ball movement can provide false feedback on lung function progress.
Breathing Trainers & Monitors Market Context
Market Overview
The breathing trainer market is segmented between simple incentive spirometers for post-operative care and more complex electronic devices for chronic respiratory management. Flow-oriented models like this one dominate the post-surgical recovery segment due to their simplicity, low cost, and effectiveness in preventing atelectasis.
Common Issues
Common problems include flimsy construction leading to air leaks, unclear measurement markings that frustrate patients, and mouthpieces that are uncomfortable or difficult to clean properly. Some cheaper models have balls that stick in chambers, providing inaccurate feedback on inhalation effort.
Quality Indicators
Look for devices with independently moving balls in separate chambers (not connected rods), a visible goal indicator line, and a mouthpiece that accommodates both nose and mouth breathing. Medical device registration or clear compliance with FDA guidelines for Class II medical devices is another key quality marker.
Review Authenticity Insights
Grade B Interpretation
A Grade B authenticity rating with an 11% estimated fake review rate indicates generally trustworthy reviews with some minimal manipulation. For a medical device like this, the high proportion of genuine reviews discussing specific recovery scenarios (like post-CABG surgery or pneumonia recovery) adds credibility.
Trust Recommendation
Focus on reviews that mention specific medical contexts like 'after my thoracotomy' or 'during COVID recovery,' as these demonstrate authentic use. Be slightly more skeptical of reviews that only mention generic 'great product' language without medical context, as these may represent the estimated 11% less authentic feedback.
Tips for Reading Reviews
Prioritize reviews that detail progression metrics ('I moved from the blue to the green ball in three days') or mention coordination with physical therapists. These specifics indicate genuine therapeutic use rather than superficial evaluation.
Expert Perspective
This incentive spirometer's exceptional 4.89/5 rating (adjusted to 4.60/5) from nearly 1,500 reviews suggests strong user satisfaction, particularly for its intended post-surgical application. The high rating maintained even after adjusting for suspicious reviews indicates genuine appreciation for its functionality in recovery protocols. The detailed authentic reviews consistently praise its role in preventing pneumonia and improving lung expansion, which aligns with clinical goals for these devices.
Purchase Considerations
Consider whether your medical provider has specified a flow-oriented device (like this model) versus a volume-oriented model. Also evaluate if you need the visual feedback of three separate chambers or if a simpler single-chamber device would suffice for your recovery timeline and motivation needs.
Comparing Alternatives
Shoppers should compare this flow-oriented design with volume-oriented spirometers and electronic models that provide digital tracking, as different conditions may benefit from different measurement approaches.